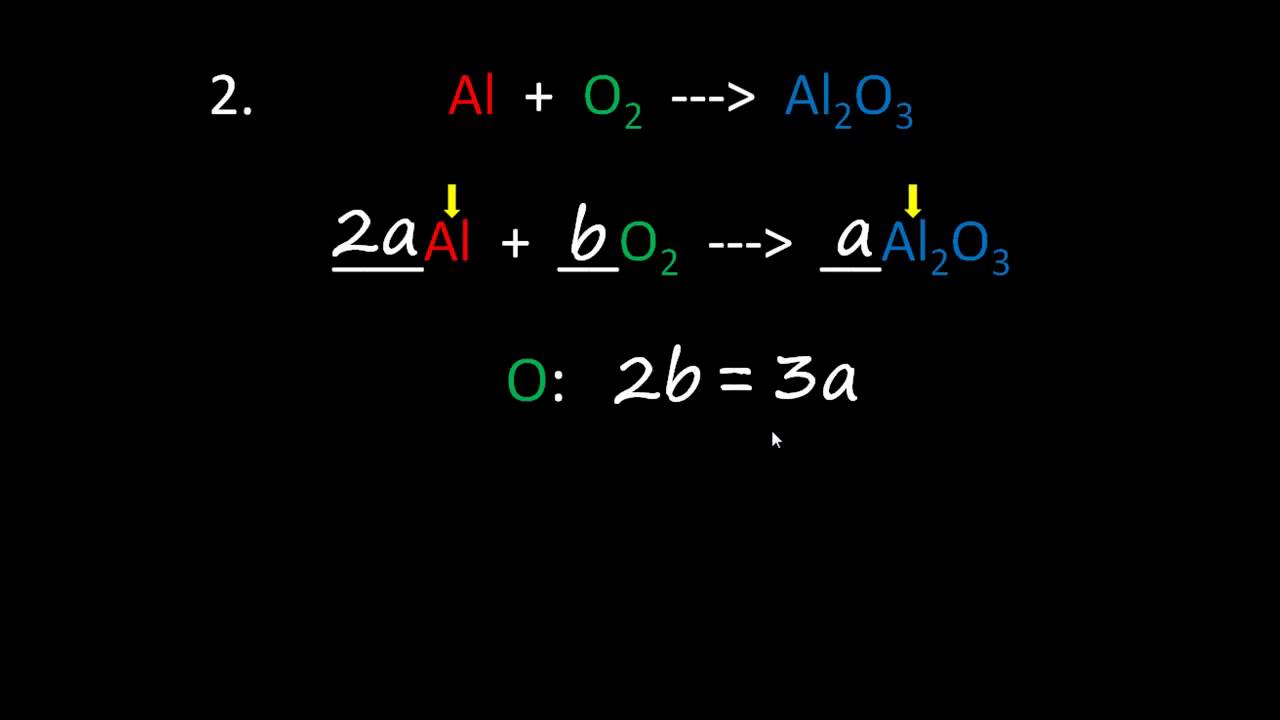
Next, applying the Conservation of Mass, which tell us that the total number of atoms of each element must be the same on both sides, write algebraic rules for each element. 
_KMnO 4 + _HCl → _MnCl 2 + _KCl + _Cl 2 + _H 2Oįirst thing we do is give each compound a letter coefficient:ĪKMnO 4 + bHCl → cMnCl 2 + dKCl + eCl 2 + fH 2O That makes absolutely no sense I’m sure without an example, so here’s an equation to balance using this strategy: Substitute the values into the other rules to obtain the balancing coefficients.Substitute and simplify to obtain a rule that equates only two letter coefficients that you can solve.Write algebraic expressions or rules for each element that equate its atoms on the LHS and RHS.Write a different letter coefficient in front of each compound in the equation.The strategy for balancing chemical equations algebraically is as follows: Simple algebraic method for balancing chemical equationsĪlgebraic method for balancing chemical equations.Algebraic method for balancing chemical equations.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
